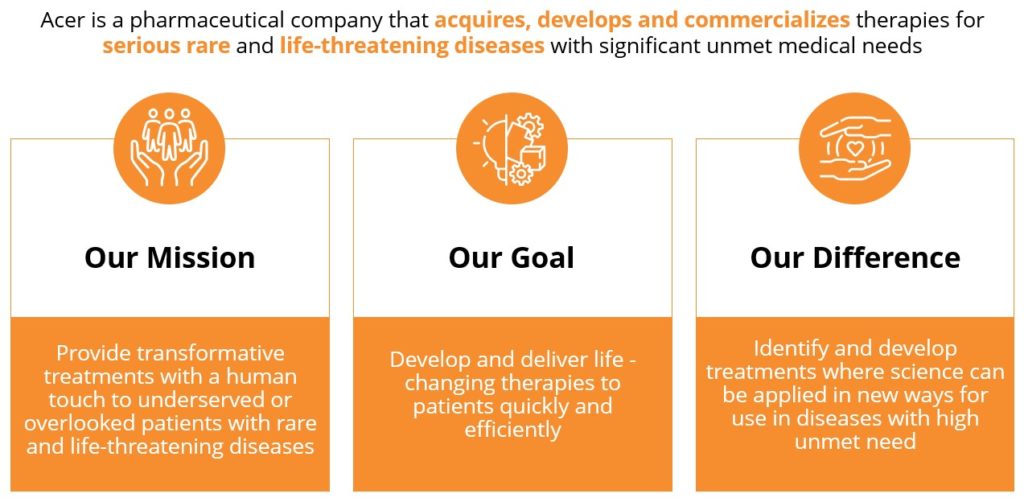
Acer is a pharmaceutical company focused on the acquisition, development and commercialization of therapies for serious rare and life-threatening diseases with significant unmet medical needs. In the U.S., OLPRUVA™ (sodium phenylbutyrate) is approved for the treatment of urea cycle disorders (UCDs) involving deficiencies of carbamylphosphate synthetase (CPS), ornithine transcarbamylase (OTC), or argininosuccinic acid synthetase (AS). Acer is also advancing a pipeline of investigational product candidates for rare and life-threatening diseases, including: OLPRUVA™ (sodium phenylbutyrate) for treatment of various disorders, including Maple Syrup Urine Disease (MSUD); and EDSIVO™ (celiprolol) for treatment of vascular Ehlers-Danlos syndrome (vEDS) in patients with a confirmed type III collagen (COL3A1) mutation.
Acer Partners