NEWTON, MA – March 27, 2023 – Acer Therapeutics Inc. (Nasdaq: ACER), a pharmaceutical company focused on the acquisition, development and commercialization of therapies for serious, rare and life-threatening diseases with significant unmet medical needs, today reported financial results for Q4 and full year ended December 31, 2022, and provided an update on the Company’s recent corporate developments.
“2022 marked Acer’s first U.S. Food and Drug Administration (FDA) product approval, for OLPRUVA™ (sodium phenylbutyrate), an innovative and responsibly priced treatment option for certain people with urea cycle disorders (UCDs) involving deficiencies of carbamylphosphate synthetase (CPS), ornithine transcarbamylase (OTC), or argininosuccinic acid synthetase (AS),” commented Chris Schelling, CEO and Founder of Acer. “We remain committed to our mission to provide transformative treatments to underserved and overlooked patients with rare and life-threatening diseases and will continue to focus our resources on launch preparation for OLPRUVA™ as well as continued pivotal Phase 3 clinical trial evaluation of EDSIVO™ for vEDS, both of which are subject to additional capital.”
Q4 2022 and Recent Highlights
- OLPRUVA™ (sodium phenylbutyrate) for oral suspension
- In October 2022, Acer was issued a Notice of Allowance from the US Patent and Trademark Office (USPTO) for US patent application No. 16/624,834 (now issued as US Patent No. 11,517,547) for claims related to a kit comprising a combination therapeutic product composed of sodium phenylbutyrate or glycerol phenylbutyrate and sodium benzoate
- In December 2022, Acer announced FDA approval of OLPRUVA™ (sodium phenylbutyrate) for oral suspension, a prescription medicine used along with certain therapy, including changes in diet, for the long-term management of adults and children weighing 44 pounds (20 kg) or greater and with a body surface area (BSA) of 1.2 m2 or greater, with UCDs, involving deficiencies of CPS, OTC or AS. OLPRUVA™ is not used to treat rapid increase of ammonia in the blood (acute hyperammonemia), which can be life-threatening and requires emergency medical treatment. More information is available at www.OLPRUVA.com
- In March 2023, Acer announced an update on its OLPRUVA™ U.S. commercial launch activities, including the ongoing addition of commercial and medical affairs resources, the introduction of its OLPRUVA™ Navigator by Acer Therapeutics patient support service, a predictable and responsible price commitment, and anticipated drug availability by early July 2023 (subject to additional capital)
- In March 2023, Acer announced results from a survey designed to quantify preferences of healthcare providers for Urea Cycle Disorders (UCDs) presented at the Society for Inherited Metabolic Disorders (SIMD) Annual Meeting. The authors concluded that optimizing nitrogen-binding medications for UCD treatment to facilitate and encourage increased patient adherence through masking taste/odor and/or enhancing other aspects of the patient experience may support improved outcomes in UCDs
- EDSIVO™ (celiprolol)
- In October 2022, Acer announced that the USPTO issued a Notice of Allowance for Acer’s patent application No. 16/930,208 and subsequently issued on December 13, 2022, as US Patent #11,523,997, for claims related to certain methods of treating vascular Ehlers-Danlos syndrome (vEDS) with celiprolol
- ACER-801 (osanetant)
- In March 2023, Acer announced that topline results from Acer’s Phase 2a proof of concept clinical trial to evaluate ACER-801 (osanetant) as a potential treatment for moderate to severe Vasomotor Symptoms (VMS) associated with menopause showed that ACER-801 was safe and well-tolerated but did not achieve statistical significance when evaluating ACER-801’s ability, when compared to placebo, to decrease the frequency or severity of hot flashes in postmenopausal women. As a result, Acer is pausing the ACER-801 program until Acer has conducted a thorough review of the full data set
- Corporate
- Ended Q4 2022 with $2.3 million in cash and cash equivalents. Acer believes its cash and cash equivalents available at December 31, 2022, together with the gross proceeds of $7.0 million from a second term loan with SWK Funding LLC which closed on January 31, 2023, $4.1 million from Acer’s ATM facility subsequent to December 31, 2022, and $2.7 million from a sale of securities (including pursuant to a registered direct offering of shares of common stock and prefunded warrants) which closed on March 24, 2023, will be sufficient to fund Acer’s anticipated operating and capital requirements into the middle of Q2 2023
Anticipated Milestones (Subject to Available Capital)
- Q2 2023: Acer intends to continue to add resources to establish its commercial and medical affairs presence in the U.S. in support of its OLPRUVA™ launch expected in Q2 2023
- Q2 2023: Negotiations with the major commercial payers and state Medicaid organizations regarding access for OLPRUVA™ are ongoing, and representatives from Acer’s OLPRUVA™ Navigator by Acer Therapeutics patient support service are expected to begin accepting prescriptions in late Q2 2023
- Q2 2023: Acer expects OLPRUVA™ wholesale acquisition cost (WAC) pricing to be listed publicly in Q2 2023
- Early July 2023: Acer anticipates OLPRUVA™ drug availability in early July 2023
- Q4 2023: Acer anticipates completing enrollment in its ongoing, pivotal Phase 3 DiSCOVER trial of EDSIVO™ (celiprolol) in patients with COL3A1-positive vEDS. The double-blind portion of the DiSCOVER trial is designed to end if statistical significance is reached at an interim analysis which occurs at accrual of 28 vEDS-related events, estimated to occur as early as approximately 18 months after completion of full enrollment, or after accrual of 46 vEDS-related clinical events
- Acer intends to explore additional lifecycle opportunities for OLPRUVA™ (sodium phenylbutyrate) in various disorders where proof of concept data exists, including in Maple Syrup Urine Disease (MSUD), Pyruvate Dehydrogenase Complex Deficiency (PCDC), rare pediatric epilepsies and various liver disorders
Q4 and Full Year 2022 Financial Results
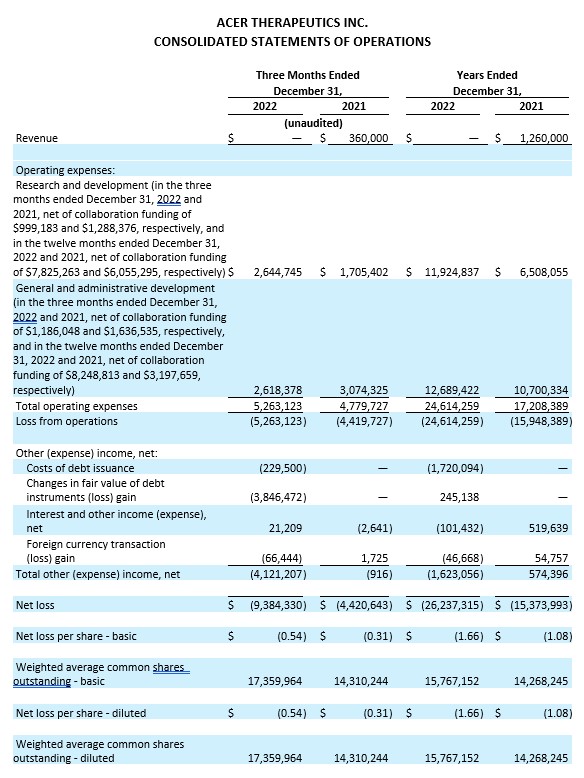
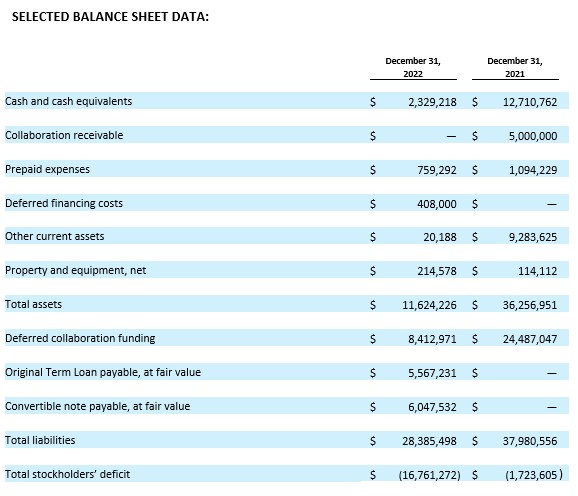
Cash Position. Cash and cash equivalents were $2.3 million as of December 31, 2022, compared to $12.7 million as of December 31, 2021. Acer believes its cash and cash equivalents as of December 31, 2022, together with the gross proceeds of $7.0 million from a second term loan with SWK Funding LLC which closed on January 31, 2023, $4.1 million from Acer’s ATM facility subsequent to December 31, 2022, and $2.7 million from a sale of securities (including pursuant to a registered direct offering of shares of common stock and prefunded warrants) which closed on March 24, 2023, will be sufficient to fund its currently anticipated operating and capital requirements into the middle of Q2 2023.
Research and Development Expenses. Research and development expenses were $2.6 million, net of collaboration funding of $1.0 million, for the three months ended December 31, 2022, as compared to $1.7 million, net of collaboration funding of $1.3 million, for the three months ended December 31, 2021. Research and development expenses for the three months ended December 31, 2022 were comprised of $1.1 million related to ACER-001, offset by $1.0 million of collaboration funding; $1.2 million related to ACER-801; $1.0 million related to EDSIVO™; and $0.3 million related to other development activities. Research and development expenses were $11.9 million, net of collaboration funding of $7.8 million, for the year ended December 31, 2022, as compared to $6.5 million, net of collaboration funding of $6.1 million, for the year ended December 31, 2021. This increase of $5.4 million was primarily due to increases in employee-related expenses and expenses related to clinical studies, partially offset by the increase in recognition of contra-expense from the collaboration funding from Acer’s Collaboration and License Agreement with Relief Therapeutics Holding AG (Relief) for the development and commercialization of OLPRUVA™ (Collaboration Agreement).
General and Administrative Expenses. General and administrative expenses were $2.6 million, net of collaboration funding of $1.2 million, for the three months ended December 31, 2022, as compared to $3.1 million, net of collaboration funding of $1.6 million, for the three months ended December 31, 2021. General and administrative expenses were $12.7 million, net of collaboration funding of $8.2 million, for the year ended December 31, 2022, as compared to $10.7 million, net of collaboration funding of $3.2 million, for the year ended December 31, 2021. This increase of $2.0 million was primarily due to increases in precommercial activities, employee-related expenses, professional services, as well as information technology, partially offset by the increase in the recognition of contra-expense from the collaboration funding from the Collaboration Agreement with Relief.
Net Loss. Net loss for the three months ended December 31, 2022 was $9.4 million, or $0.54 net loss per share (basic and diluted), compared to a net loss of $4.4 million, or $0.31 net loss per share (basic and diluted), for the three months ended December 31, 2021. Net loss for the year ended December 31, 2022 was $26.2 million, or $1.66 net loss per share (basic and diluted), compared to a net loss of $15.4 million, or $1.08 net loss per share (basic and diluted), for the year ended December 31, 2021.
For additional information, please see Acer’s Annual Report on Form 10-K filed today with the SEC.
About Acer Therapeutics
Acer is a pharmaceutical company focused on the acquisition, development and commercialization of therapies for serious rare and life-threatening diseases with significant unmet medical needs. In the U.S., OLPRUVA™ (sodium phenylbutyrate) is approved for the treatment of urea cycle disorders (UCDs) involving deficiencies of carbamylphosphate synthetase (CPS), ornithine transcarbamylase (OTC), or argininosuccinic acid synthetase (AS). Acer is also advancing a pipeline of investigational product candidates for rare and life-threatening diseases, including: OLPRUVA™ (sodium phenylbutyrate) for treatment of various disorders, including Maple Syrup Urine Disease (MSUD); EDSIVO™ (celiprolol) for treatment of vascular Ehlers-Danlos syndrome (vEDS) in patients with a confirmed type III collagen (COL3A1) mutation; and ACER-801 (osanetant) for treatment of Vasomotor Symptoms (VMS), post-traumatic stress disorder (PTSD) and prostate cancer. For more information, visit www.www.acertx.com.
Acer Forward-Looking Statements This press release contains “forward-looking statements” that involve substantial risks and uncertainties for purposes of the safe harbor provided by the Private Securities Litigation Reform Act of 1995. All statements, other than statements of historical facts, included in this press release are forward-looking statements. Examples of such statements include, but are not limited to, statements about plans for the commercialization of OLPRUVA™ for oral suspension in the U.S. for the treatment of certain patients with UCDs involving deficiencies of CPS, OTC, or AS, including negotiations with commercial payers and Medicaid organizations regarding access as well as the timing of drug availability and the expected commercial launch, statements about plans and potential milestones for the continued clinical development of OLPRUVA™ in other indications, statements about plans and potential milestones for the continued clinical development of EDSIVO™ for treatment of vEDS in patients with a confirmed type III collagen (COL3A1) mutation, statements about plans for the development of ACER-801, statements about our anticipated 2023 milestones, and statements about our capital requirements and sufficiency and duration of our current cash and cash equivalents. Our efforts to commercialize OLPRUVA™ for oral suspension in the U.S. for the treatment of certain patients with UCDs involving deficiencies of CPS, OTC, or AS are at an early stage, we currently do not have fully developed marketing, sales or distribution capabilities, and there is no guarantee that we will be successful in our commercialization efforts. Our pipeline products (including OLPRUVA™ for indications other than UCDs as well as EDSIVO™ and ACER-801) are under investigation and their safety and efficacy have not been established and there is no guarantee that any of our investigational products in development will receive health authority approval or become commercially available for the uses being investigated. We may not actually achieve the plans, carry out the intentions or meet the expectations or projections disclosed in the forward-looking statements and you should not place undue reliance on these forward-looking statements. Such statements are based on management’s current expectations and involve risks and uncertainties. Actual results and performance could differ materially from those projected in the forward-looking statements as a result of many factors, including, without limitation, the availability of financing to fund our commercialization efforts, our pipeline product development programs and our general corporate operations as well as risks related to drug development and the regulatory approval process, including the timing and requirements of regulatory actions. We disclaim any intent or obligation to update these forward-looking statements to reflect events or circumstances that exist after the date on which they were made. You should review additional disclosures we make in our filings with the Securities and Exchange Commission, including our Annual Report on Form 10-K and Quarterly Reports on Form 10-Q. You may access these documents for no charge at http://www.sec.gov.
Corporate and IR Contact
Jim DeNike
Acer Therapeutics Inc.
[email protected]
+1-844-902-6100
Nick Colangelo
Gilmartin Group
[email protected]
+1-332-895-3226