NEWTON, MA – Nov. 10, 2020 – Acer Therapeutics Inc. (Nasdaq: ACER), a pharmaceutical company focused on the acquisition, development and commercialization of therapies for serious rare and life-threatening diseases with significant unmet medical needs, today reported financial results for the third quarter ended September 30, 2020, and provided an update on the Company’s recent corporate developments.
“This quarter, we continued to make important progress in the advancement of our four programs,” said Chris Schelling, CEO and Founder of Acer. “We received valuable feedback from the FDA regarding our regulatory strategy for ACER-001 for urea cycle disorders patients, and assuming all our development activities stay on track, we anticipate having a pre-NDA meeting in the mid-first half of 2021. Regarding our emetine program, several in vivo studies are currently being conducted to evaluate the drug’s safety and its specific activity against SARS-CoV-2 in the Syrian Golden hamster and ferret models. Coupled with the drug’s in vitro data and emetine’s long history of use in humans for other indications, we should have a good understanding of this candidate’s potential as a therapeutic against COVID-19 relatively soon.”
Third Quarter 2020 and Recent Highlights
- Emetine
- Animal studies ongoing to further evaluate emetine’s safety and antiviral activity against COVID-19, the disease caused by infection with the SARS-CoV-2 virus
- Obtained additional pre-Investigational New Drug (IND) feedback from the Division of Antivirals (DAV) at the U.S. Food and Drug Administration (FDA)
- Progressing toward U.S.-based cGMP synthetic active pharmaceutical ingredient (API) manufacturing in 2021
- Continued to work with federal agencies and private research organizations toward the goal of securing non-dilutive funding in support of emetine development
- ACER-001
- Received feedback on the Type C meeting with the FDA regarding requirements for the planned ACER-001 505(b)(2) New Drug Application (NDA) submission for treatment of urea cycle disorders (UCDs)
- Planning to conduct a single-center, single-blind, randomized, single-dose crossover trial designed to demonstrate bioequivalence (BE) of ACER-001 compared to BUPHENYL® under fed conditions, in approximately 36 healthy adults
- Gained insight from market research with key stakeholders – geneticists, dietitians and UCDs patients – to better understand the reaction to ACER-001’s value proposition and begin quantifying the potential market opportunity in UCDs
- EDSIVO™
- Developing a plan to generate confirmatory evidence needed to support the potential resubmission of the EDSIVO™ NDA (although neither EDSIVO™ NDA resubmission nor approval is assured)
- Osanetant
- Continued to advance active pharmaceutical ingredient (API) manufacturing and other necessary steps to support IND submission
- Corporate
- Ended the third quarter with $6.2 million in cash and cash equivalents. Acer believes its cash position at the end of the third quarter, combined with an additional $1.0 million of proceeds subsequently received through October 26, 2020 from the sales of common stock under its ATM facility and through its equity purchase agreement with Lincoln Park Capital, will be sufficient to fund its current operations into the first quarter of 2021, excluding support for the planned emetine Phase 2/3 clinical trial, which is also subject to ongoing discussions with the FDA
Upcoming Milestones
- Emetine
- Anticipate in vivo efficacy results from ongoing animal studies evaluating emetine’s anti-viral activity against COVID-19 by the end of the fourth quarter 2020
- Targeting IND submission and potential initiation of the planned Phase 2/3 trial evaluating emetine in high-risk COVID-19 outpatients in the first half of 2021, subject to ongoing discussions with the FDA and additional capital
- ACER-001
- Targeting completion of a BE trial of ACER-001 compared to BUPHENYL® under fed conditions, in approximately 36 healthy adults, in the first quarter of 2021
- Anticipate holding a pre-NDA meeting with FDA in mid-first half of 2021, assuming successful and timely completion of the ongoing development activities (including the planned BE fed trial, nonclinical work and evaluation of long-term product stability data) and subject to additional capital
- EDSIVO™
- Expect to submit a meeting request with FDA to discuss Acer’s proposed plan to provide sufficient confirmatory evidence by the end of the fourth quarter of 2020
- If successful, potentially satisfy the substantial evidence of effectiveness needed to support a possible resubmission of the EDSIVO™ NDA (although neither EDSIVO™ NDA resubmission nor approval is assured)
- Osanetant
- Targeting IND submission in the second quarter of 2021 subject to discussions with FDA
- Plan to initiate a Phase 1/2 pharmacokinetic/pharmocodynamic and safety trial evaluating osanetant in patients with medically and/or surgically induced vasomotor symptoms (iVMS) in the second half of 2021, subject to additional capital
Financial Results for the Third Quarter 2020
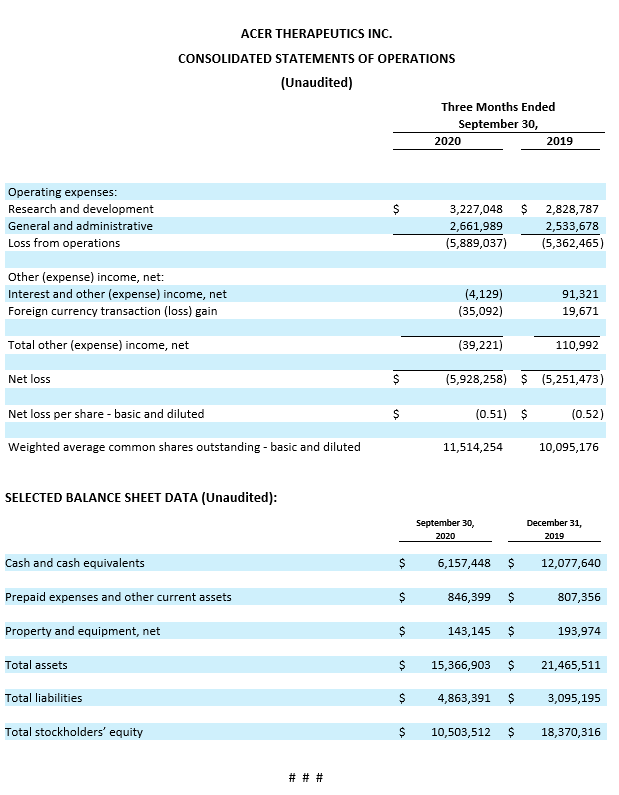
Cash position. Cash and cash equivalents were $6.2 million as of September 30, 2020, compared to $12.1 million as of December 31, 2019. Acer believes its cash position at the end of the third quarter, combined with an additional $1.0 million of proceeds subsequently received through October 26, 2020 from the sales of common stock under its ATM facility and through its purchase agreement with Lincoln Park Capital, will be sufficient to fund its current operations into the first quarter of 2021, excluding support for the planned emetine Phase 2/3 clinical trial, which is also subject to ongoing discussions with the FDA.
Research and Development Expenses. Research and development expenses were $3.2 million for the three months ended September 30, 2020, compared to $2.8 million for the three months ended September 30, 2019. This increase of $0.4 million was primarily due to an increase in expenses for contract research related to preclinical studies, partially offset by decreases in regulatory consulting and contract manufacturing expenses. Research and development expenses for the three months ended September 30, 2020 were primarily comprised of $1.8 million related to emetine, $1.1 million related to ACER-001, and $0.3 million related to osanetant.
General and Administrative Expenses. General and administrative expenses were $2.7 million for the three months ended September 30, 2020, compared to $2.5 million for the three months ended September 30, 2019. This increase of $0.2 million was primarily due to an increase in expenses for consulting and professional services, partially offset by decreases in expenses related to travel and information technology.
Net Loss. Net loss for the three months ended September 30, 2020 was $5.9 million, or $0.51 net loss per share (basic and diluted), compared to a net loss of $5.3 million, or $0.52 net loss per share (basic and diluted), for the three months ended September 30, 2019.
For additional information, please see Acer’s Quarterly Report on Form 10-Q filed today with the SEC.
About Acer Therapeutics Inc.
Acer is a pharmaceutical company focused on the acquisition, development and commercialization of therapies for serious rare and life-threatening diseases with significant unmet medical needs. Acer’s pipeline includes four programs: emetine hydrochloride for the treatment of patients with COVID-19; ACER-001 (a taste-masked, immediate release formulation of sodium phenylbutyrate) for the treatment of various inborn errors of metabolism, including urea cycle disorders (UCDs) and Maple Syrup Urine Disease (MSUD); EDSIVO™ (celiprolol) for the treatment of vascular Ehlers-Danlos syndrome (vEDS) in patients with a confirmed type III collagen (COL3A1) mutation; and osanetant for the treatment of induced Vasomotor Symptoms (iVMS). Each of Acer’s product candidates is believed to present a comparatively de-risked profile, having one or more of a favorable safety profile, clinical proof-of-concept data, mechanistic differentiation and/or accelerated paths for development through specific programs and procedures established by the FDA. For more information, visit www.www.acertx.com.
Forward-Looking Statements
This press release contains “forward-looking statements” that involve substantial risks and uncertainties for purposes of the safe harbor provided by the Private Securities Litigation Reform Act of 1995. All statements, other than statements of historical facts, included in this press release regarding strategy, future operations, timelines, future financial position, future revenues, projected expenses, regulatory submissions, actions or approvals, cash position, liquidity, prospects, plans and objectives of management are forward-looking statements. Examples of such statements include, but are not limited to, statements relating to the potential for our product candidates to safely and effectively treat diseases and to be approved for marketing; the commercial or market opportunity of any of our product candidates in any target indication and any territory; our ability to secure the additional capital necessary to fund our various product candidate development programs; the adequacy of our capital to support our future operations and our ability to successfully fund, initiate and complete clinical trials and regulatory submissions; the ability to protect our intellectual property rights; our strategy and business focus; and the development, expected timeline and commercial potential of any of our product candidates. We may not actually achieve the plans, carry out the intentions or meet the expectations or projections disclosed in the forward-looking statements and you should not place undue reliance on these forward-looking statements. Such statements are based on management’s current expectations and involve risks and uncertainties. Actual results and performance could differ materially from those projected in the forward-looking statements as a result of many factors, including, without limitation, risks and uncertainties associated with the ability to project future cash utilization and reserves needed for contingent future liabilities and business operations, the availability of sufficient resources to fund our various product candidate development programs and to meet our business objectives and operational requirements, the fact that the results of earlier studies and trials may not be predictive of future clinical trial results, the protection and market exclusivity provided by our intellectual property, the substantial costs and diversion of management’s attention and resources which could result from pending securities litigation, risks related to the drug development and the regulatory approval process, including the timing and requirements of regulatory actions, and the impact of competitive products and technological changes. We disclaim any intent or obligation to update these forward-looking statements to reflect events or circumstances that exist after the date on which they were made. You should review additional disclosures we make in our filings with the Securities and Exchange Commission, including our Quarterly Reports on Form 10-Q and our Annual Report on Form 10-K. You may access these documents for no charge at http://www.sec.gov.
Investor Contact:
Hans Vitzthum
LifeSci Advisors
Ph: 617-430-7578
[email protected]
Jim DeNike
Acer Therapeutics Inc.
Ph: 844-902-6100
[email protected]